THE SPECTRA OF COMETS
Introduction
- Linear 2000WM
- 2001Q4 (Neat)
- Machholz 2004Q2
Introduction (spectrum of comet obtained in your kitchen)
G. B. Donati in 1864 from Florence was the first astronomer who observed the spectrum
of a comet. He found three emission bands in the blue, green and yellow overlapped to
the continuum of the solar spectrum.
Four years later, Huggins observed the same bands in the comet Tempel I and was able to
identify these bands with those observed by Swan in the candle flame and in the sparkle
excitation of ethylic alcohol. Swan bands can also be observed easily in the flame of the
gas burner of our kitchens (see figure 1).
Nowadays we know that these bands are emitted by the radical C2 that is
continuosly produced by dissociation of hydrocarbons and continuously transformed
into stable CO2. Such a short lived molecules are called "radicals".
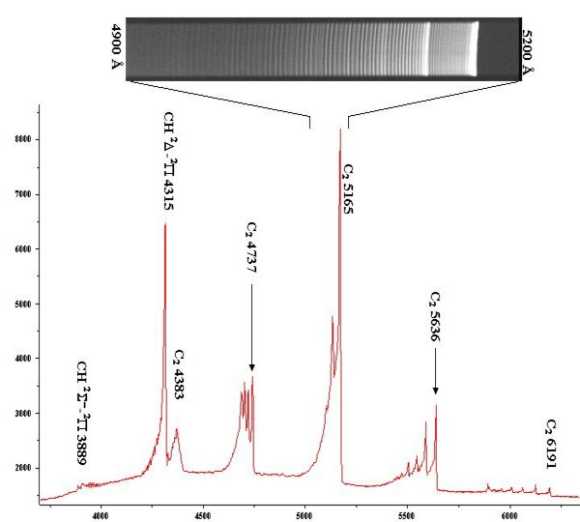
|
Picture 1: Spectrum of the propane gas flame (but also methane gives the same C2
and CH bands) recorded with 2.5 Ĺ resolution.
In the inset it is shown a magnification of a small portion of the spectrum,
recorded with 0.8 Ĺ resolution, that shows the roto-vibrational sublevels of each
electronic transition. Compare this spectrum with those obtained for real comets!
|
In comets spectra other bands are produced also by radicals CN, C3, CH,
NH2, OH that are continuously produced by the photodissociation of more
complex molecules.
In the spectra of comets we can also find the spectral signature of some ions (that have the same origin of radicals) like CO+,
CH+, OH+, NH2+.
Hydrogen (that is of course a major compound of comets) cannot be observed in the visible
range because at low temperature (like in space surrounding comets) electrons cannot
jump so high to produce the Balmer series (that originates from n>2 states to n=2
state). Thus, only the Lyman series, originating from the lower energy levels
(from n>1 to n=1 fundamental state), can be observed from space in the UV range.
Goto observed comets spectra:
Linear 2000 WM
- 2001Q4 (Neat)
- Macholz 2004Q2
|
|

